Materialization of straw is one of the important research directions in the utilization of agricultural waste. Adhesives are usually needed to bond straw to form a certain shape. The most widely used adhesive is urea-formaldehyde resin adhesive, which has the advantages of simple manufacturing process, convenient use, low cost and good performance. The development of new functions of urea-formaldehyde-based materials plays an important role in promoting and supporting the development of straw materials. In addition to being used as adhesives, urea-formaldehyde-based materials can be used as slow-release fertilizers on crops, and they can also be used as adsorbent materials in environmental fields.
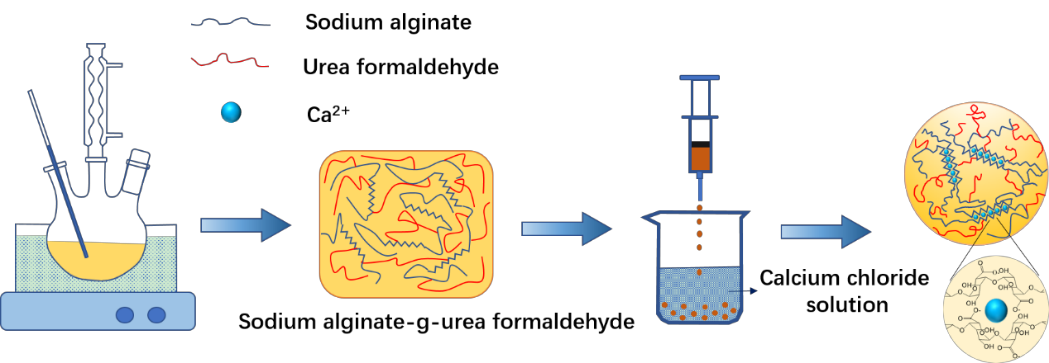
Recently, the agricultural waste recycling research team of the Circular Agriculture Research Center cooperated with the University of Florida, Urea formaldehyde (UF) was grafted onto the backbone of alginate to prepare microbeads as an adsorbent for the removal of heavy metal ions from aqueous solutions. The expensive alginate was crosslinked with cheaper UF at different ratios (1: 2.5∼1: 12.5) to produce sturdy alginate-UF beads at lower cost. Characterization results showed that UF modification enhanced the pore network and structural stability of the beads, which can be attributed to the reduced intermolecular forces and plentiful of nitrogen and oxygen donor atoms of the beads. The swelling of air-dried alginate-UF beads in different solutions was much lower than that of the unmodified alginate beads, confirming the improved stability. The adsorption capacities of Pb2+, Cd2+, and Cu2+ on air-dried alginate-UF (1: 2.5) beads were 39.88%, 9.29%, and 9.52% higher than those of the corresponding unmodified alginate beads, respectively. The adsorption of heavy metals on the alginate-UF beads was mainly controlled by ion exchange, complexation, and electrostatic interaction mechanisms. Relevant research results were published in Journal of Hazardous Materials (IF: 9.038).
A simple foaming method was applied to fabricate urea formaldehyde (UF) microspheres with crosslinked porous structures for environmental remediation of heavy metals. All the foamed UF microspheres showed good removal of heavy metals ions (Pb(II), Cu(II), and Cd(II)) in both single and mixed-metal solutions. Further investigations of Pb(II) adsorption on a selected UF microspheres showed fast kinetics and relatively high adsorption capacity (21.5 mg/g), which can be attributed to the meso-porous structure and abundance of oxygen surface functional groups of the microspheres. Both experimental and model results showed that chelation or complexation interactions between Pb(II) and the surface functional groups were responsible to the strong adsorption of the heavy metal ions on the microspheres. Hydrochloric acid (0.05 M) successfully desorbed Pb(II) from the post-adsorption micro-spheres for multiple times and the regenerated microspheres showed high Pb(II) removal rates (>96%) in five adsorption-desorption cycles. With many promising advantages, foamed UF microspheres show great potential as a wastewater treatment agent for heavy metal removal. Relevant research results were published in Chemosphere (IF: 5.778).
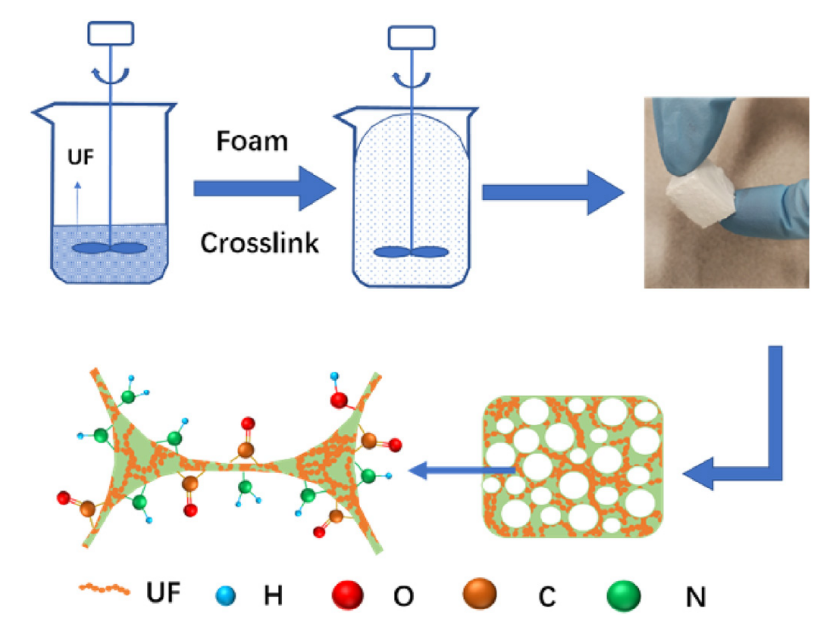
The first author of both papers is associate professor Ping Qu. This research was funded by the National Natural Science Foundation of China Youth Fund (11605077), China Scholarship Counsil (201808320076) and Jiangsu Independent Innovation Fund (CX (192003)).
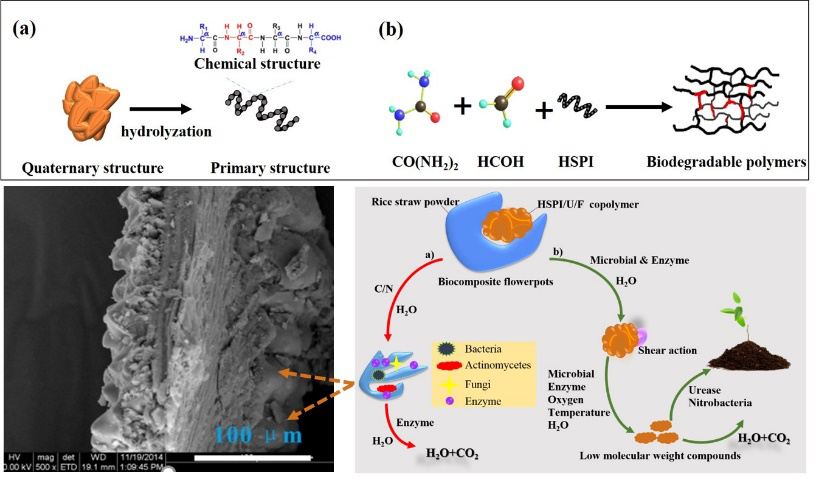
In addition, the team members used modified urea-formaldehyde resin as the matrix and straw fibers as reinforcement materials to prepare flower pots. Molecular structure, morphology, biodegradability and microbial population of the resulting materials were investigated. FTIR and SEM results indicated that the hydrolyzed soybean protein isolate/urea/formaldehyde resin have been successfully prepared. The prepared biocomposite flowerpots showed good biodegradation, and the introduction of hydrolyzed soybean protein isolate increased the degradation rate of biocomposite flowerpots, which was nearly 50% at 24 months. After 30 days of controlled composting, the CO2 release accumulation of biocomposite flowerpots could reach 24 g. During the process of biocomposite flowerpots degradation, the number of bacteria and fungi on the surface was higher than that of actinomyces, indicating the bacteria and fungi selectively accumulated on the biocomposite flowerpots composites surface to accelerate its degradation. At the same time, nitrate ion could also be formed by nitrobacteria, which promoted plant growth. There is no doubt that the obtained biodegradable copolymer possessed great potential for biocomposite flowerpots application. Relevant research results were published in Composites Part B: Engineering (IF: 7.635). Associate professor Enhui Sun is the first author, and professor Huang Hongying is the corresponding author.
The team also developed a new type of slow-release fertilizer by blocking the hydrolyzed protein in the urea-formaldehyde molecular chain. Urea formaldehyde is one of the great potential slow release nitrogen fertilizers. However, low molecular weight of urea formaldehyde releases nitrogen faster in the early growing season, while high molecular weight of urea formaldehyde releases nitrogen slower during the late growing season. We developed an effective method to optimize the nitrogen release rate in urea formaldehyde by copolymerizing with other materials. The nitrogen release rate was improved by the introduced hydrolyzed soy protein. The nitrogen release rate of modified urea formaldehyde was reduced in the earlier stage and faster in the later stage from soil column leaching experiments. The hydrolysis degree of soy protein was lower, the nitrogen release rate was faster. The nitrogen release rate of the modified urea formaldehyde was up to 74.4%, and the nitrogen release rate of unmodified urea formaldehyde was 46.8%, with the nitrogen release rate was improved 58.9%. Fourier transform infrared spectroscopy, X-ray diffraction, thermogravimetric analysis, scanning electron microscope were applied to characterize the hydrolyzed soy protein modified urea formaldehyde. The functional groups of soy protein were exposed after hydrolyzation. The hydrolysis degree of soy protein became higher, more formaldehyde was reacted. The reactive groups of hydrolyzed soy protein can react with formaldehyde and copolymerize into the urea formaldehyde molecular. The crystallinity of modified urea formaldehyde was lower because of the introduced hydrolyzed soy protein. The maximum mass loss rate of temperature of modified urea formaldehyde decreased as the decreasing of the hydrolysis degree of soy protein, which decreased 11.9 ºC at most, compared with non-modified urea formaldehyde. Therefore, the nitrogen release rate can be regulated by the hydrolysis degree of soy protein to adapt to crop life cycles, which was an effective way to improve nitrogen use efficiency. The hydrolyzed soy protein modified urea formaldehyde shows a good application potential. Relevant research results were published in Transactions of the Chinese society of Agricultural Engineering (EI). Associate professor Ping Qu is the first author, and professor Hongying Huang is the corresponding author.
Paper link:
(1)Journal of Hazardous Materials
https://doi.org/10.1016/j.jhazmat.2020.122664
(2)Chemosphere
https://doi.org/10.1016/j.chemosphere.2019.125004
(3)Composites Part B: Engineering
https://doi.org/10.1016/j.compositesb.2018.11.121
urea-formaldehyde resin other papers related links:
(4)Transactions of the Chinese society of Agricultural Engineering
DOI: 10.11975/j.issn.1002-6819.2016.21.033
(5)Transactions of the Chinese society of Agricultural Engineering
DOI: 10.3969/j.issn.1002-6819.2014.13.028



