The fast development of agriculture and livestock husbandry produces nearly 5.0 billion tons of crop straw and animal manure annually in China. Aerobic composting is an economic and effective way to dispose and recycle organic solid wastes. Organic matter (OM) is the major metabolizable component in the raw materials that can be transformed into stable humic substance (HS) through the action of microorganisms. Lignocellulose is the most stable macromolecular OM in organic solid waste, and its degradation and transformation is closely related to the humification and stabilization process of aerobic composting. During the traditional aerobic composting process, the high content of lignocellulose and low humification degree of OM lead to low conversion efficiency of OM and poor product quality.
In order to solve this problem, the agricultural input pollution reduction and control team of the Institute of Resources and Environmental Sciences carried out the research and development of microbial enhanced humification technology. In this study, we examine the effect of thermal pretreatment (TP) on lignocellulose degradation, humification and bacterial community succession during composting, identify the key environmental factors that influence the bacterial community shift, and reveal the key microorganisms involved in lignocellulose transformation. In addition, the functional microorganisms involved in lignocellulose humification were successfully isolated and their transformation mechanism was studied.

Figure 1
Our results indicated that TP accelerated the composting process and promoted the lignocellulose decomposition and HS formation. The degradation degree of total organic carbon (TOC) increased by 100% after TP. The degradation rate of cellulose, hemicellulose and lignin increased by 78%, 10% and 109%, respectively. The humus content and humification index increased by 14% and 38%, respectively. Through 16S rRNA high-throughput sequencing, we found that TP changed the initial bacterial community structure in the raw materials and affected the succession of bacterial community in the subsequent composting process (Fig. 1a). After TP, the relative abundance of thermophilic bacteria increased significantly (P < 0.05), and thermophilic bacteria represented by Thermobifida, Bacillus and Planococcus became the dominant bacterial group (Fig. 1b). Correlation analysis further showed that thermophilic bacteria, Thermobifida, Planiflum, Truepera and Thermomonospora were potentially involved in the biodegradation and humification of lignocellulosic organics (Fig. 1c-d). Canonical correspondence analysis (CCA) showed that TP significantly altered the bacterial community succession during composting by changing the main environmental factor from dissolved organic carbon (DOC) to temperature (Fig. 2). The increased relative abundance of thermophilic bacteria was responsible for the improved composting efficiency by accelerating the biodegradation and humification process of lignocellulosic fractions.
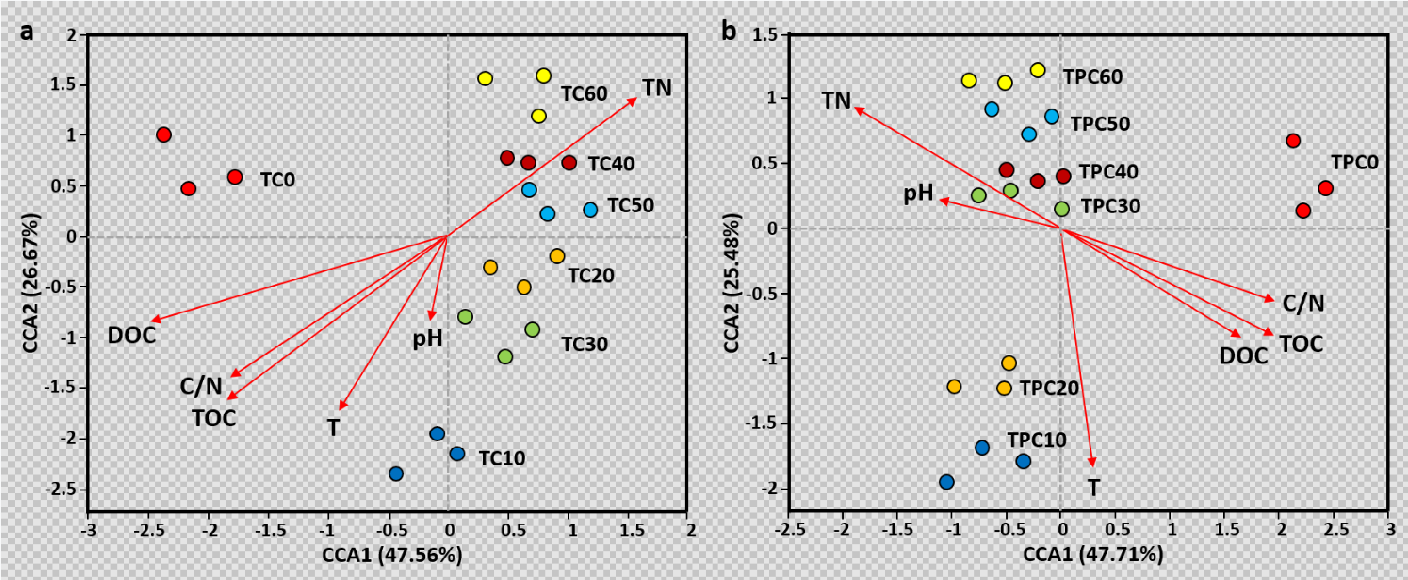
Figure 2
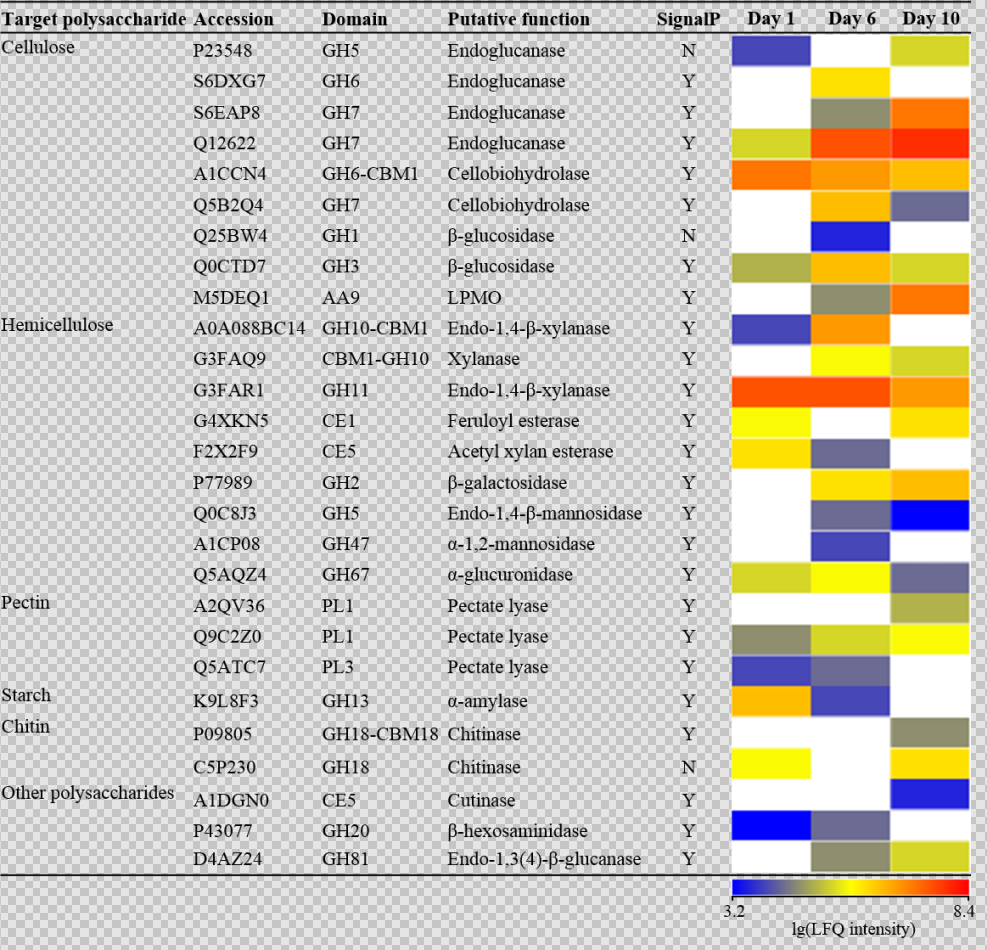
Figure 3
Our team further isolated the dominant thermophilic strain Malbranchea cinnamomea N12 from the high-temperature composting system. We employed a combination of substrate degradation assay, enzyme activity detection, hydrolysis performance test and enzyme system analysis to study its lignocellulose degradation characteristics and enzymatic mechanism. M. cinnamomea N12 could rapidly degrade lignocellulose in natural substrate at 50 ℃, and the degradation rate of cellulose and hemicellulose reached 19% and 38% within 10 days, respectively. The results showed that the hydrolysis activity of M. cinnamomea N12 was significantly higher than that of Trichoderma reesei at 40-75 ° C, and it could increase the yield of glucose and xylose of wheat straw by 32% and 166%, respectively. Quantitative proteomics was used to study the temporal dynamics of extracellular degrading enzymes of M. cinnamomea N12. M. cinnamomea N12 produced a variety of lignocellulose degrading enzymes, which displayed different secretion sequence and expression level. In the early stage of degradation, xylanase and cellobiohydrolase were highly expressed, which acted on hemicellulose and cellulose crystalline area around lignocellulose matrix; in the middle stage, the expression of endoglucanase increased, which degraded non-crystalline area of cellulose microfiber chain; in the late stage, polysaccharide monooxygenase became abundant enzyme and cleaved the β - 1,4-glycosidic bond of cellulose in crystalline region. These enzymes had different substrate specificity and worked synergistically to complete the degradation of lignocellulose. These results are of significant importance for understanding the mechanism of lignocellulose degradation by thermophilic microorganisms and constructing efficient functional microbial agents.
In summary, the above studies unveil the mechanism of thermophilic functional microorganisms promoting the biodegradation and humification of lignocellulose. Our research has important implications for the production of ecological high-quality agricultural inputs and the pollution prevention in agricultural environment. This research has been published in Bioresource Technology and Microbial Cell Factories. The first author is associate professor Ning Zhu, and the corresponding author is Professor Hongmei Jin. This work is financially supported by National Natural Science Foundation of China (51908255), Natural Science Foundation of Jiangsu Province (BK20180301) and Jiangsu Agricultural Science and Technology Innovation Fund (CX(19)3092).
Article links:
[1] Zhu, N., Gao, J., Liang D., et al. Thermal pretreatment enhances the degradation and humification of lignocellulose by stimulating thermophilic bacteria during dairy manure composting. Bioresource Technology 2021, 319:124149. (IF 7.539)
[2] Zhu, N., Jin, H., Kong, X., et al. Improving the fermentable sugar yields of wheat straw by high-temperature pre-hydrolysis with thermophilic enzymes of Malbranchea cinnamomea. Microbial Cell Factories 2020, 19:149. (IF4.187)


